お店のコメント(スペック情報を含む場合もあり)
内容説明 As the generic pharmaceutical industry continues to grow and thrive, so does the need to conduct efficient and successful bioequivalence studies. In recent years, there have been significant changes to the statistical models for evaluating bioequivalence, and advances in the analytical technology used to detect drug and metabolite levels have made bioequivalence testing more difficult to conduct and summarize. The Handbook of Bioequivalence Testing offers a complete and timely description of every aspect of bioequivalence testing, including worldwide regulatory requirements for filing for approval of generic drugs, applying for a waiver, securing regulatory approval of reports, and obtaining regulatory certification of facilities conducting bioequivalence studies.
商品ジャンル
商品名
最終調査日時
2015/11/15 (Sun) 23:27:27
価格の変動(直近3回 : ¥0は未調査回)
取得日時
販売価格
ポイント
実質価格
在庫状態
2015/11/15 (Sun) 23:27:27
¥22,614
0 %
¥22,614
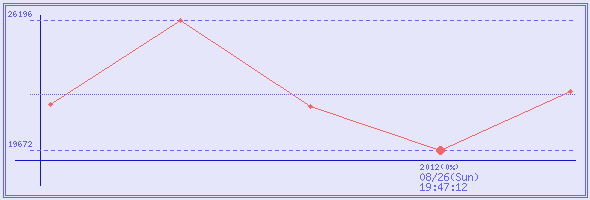
2012/08/26 (Sun) 19:47:12
¥19,672
0 %
¥19,672
2011/11/17 (Thu) 16:33:42
¥21,831
0 %
¥21,831
サイト内キーワード検索
商品名の検索は通常の商品検索ボックスで。
コメントやスペックなどから検索したい場合はこちらから。
コメントやスペックなどから検索したい場合はこちらから。
広告





